
|
Erika Tsingos |
| Research | Team | Google Scholar | Contact |
Computational Animal Development Group
Animal developmental biology is full of fascinating questions.

In our team, we address these questions using computer models. Models are great tools to check if our current understanding is consistent with expected outcomes, and to test mechanistic hypotheses for their feasibility. A well-crafted model is also predictive: In our everyday lives we all rely on weather models to predict rainfall. Our ultimate goal is to generate predictive models of animal development with all of its complexities.
Depending on the specific question and the prior biological knowledge, we craft our models using approaches such as ordinary differential equations or multiscale cell-based models using cellular Potts or center-based formalisms.
Ongoing Research
Investigating how extracellular matrix affects cell migratory phenotype
Cell migration is a fundamental process in developmental biology. It is also critical in early cancer metastasis. If we understand what causes some cells to move and others not to, we could put an early stop to cancer cell migration.
Even in genetically identical cancer cells, migration is highly heterogeneous. Some cells do not migrate at all, while others migrate individually or as a collective [1]. Structural and mechanical properties of the extracellular matrix are known to affect this migratory phenotype [2].
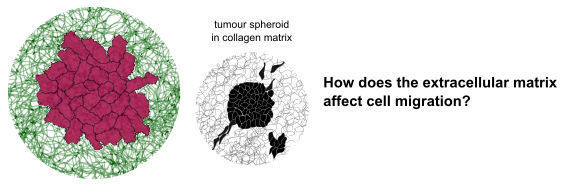
This project will build on previous work combining a cellular Potts simulation with a coarse-grained model of collagen fibers [3] to simulate a tumour spheroid embedded in a collagen matrix.
The major aims are:
- Develop a model of cell migration in fibrous matrix.
- Calibrate the model to confocal microscopy data from experimental collaborators working with tumour spheroids.
- Characterize how cell and matrix parameters affect the migratory phenotype.
References:
- Friedl, P., Locker, J., Sahai, E., & Segall, J. E. (2012). Classifying collective cancer cell invasion. Nature cell biology, 14(8), 777-783. [link]
- Boot, R. C., Koenderink, G. H., & Boukany, P. E. (2021). Spheroid mechanics and implications for cell invasion. Advances in Physics: X, 6(1), 1978316. [link]
- Tsingos, E., Bakker, B. H., Keijzer, K. A., Hupkes, H. J., & Merks, R. M. (2023). Hybrid cellular Potts and bead-spring modeling of cells in fibrous extracellular matrix. Biophysical Journal. [link]
Proliferation and cell fate specification in C. elegans
C. elegans is a small nematode worm and a fascinating model organism. One of its salient characteristics is eutely - the fact that each individual worm has a specified and fixed number of somatic cells - devoid of any randomness. Every single somatic cell division occurs at a predefined time, with a predefined division axis, and resulting in a predefined pattern of daughter cell fates. How does the worm achieve this clockwork precision?
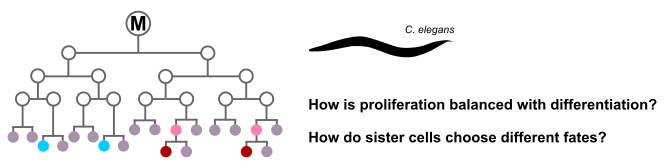
In this project we take a closer look at the C. elegans postembryonic M cell lineage. The offspring of the M cell undergo a different number of cell division rounds, and give rise to multiple cell fates. A few key mutations completely throw off the balance between proliferation and differentiation in this lineage, leading to massive overproliferation [1]. Several other mutants maintain the correct number of cell divisions, but show fate transformations [2]. Furthermore, many mutants lose their incredible precision. We will unravel the underlying control mechanisms using ordinary differential equation-based models informed by RNA sequencing data provided by collaborators.
This project is a collaboration with the Ten Tusscher group and the Van den Heuvel group.
References:
- Ruijtenberg, S., & van den Heuvel, S. (2015). G1/S inhibitors and the SWI/SNF complex control cell-cycle exit during muscle differentiation. Cell, 162(2), 300-313. [link]
- Krause, M., & Liu, J. (2012). Somatic muscle specification during embryonic and post‐embryonic development in the nematode C. elegans. Wiley Interdisciplinary Reviews: Developmental Biology, 1(2), 203-214. [link]
Development of the thymus and leukemogenesis
The vertebrate thymus is the organ where immature T-cells of the immune system differentiate into either αβ or γδ cells, and then undergo thymic selection to prevent auto-immunity. Fish model organisms allow to study the coming and going of T-cells in the living organism, giving unique insights into the turnover of thymocyte populations, while also enabling genetic manipulations [1].
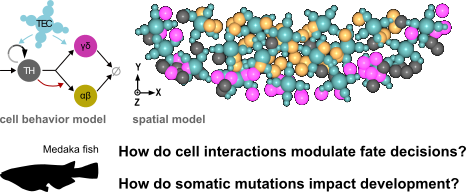
Based on quantitative fluorescence microscopy data of the Medaka (Oryzias latipes) larval thymus, we previously developed and calibrated a spatial center-based model [2]. We used this model to understand how cell-intrinsic signals and extrinsic signals emanating from thymic epithelial cells (TECs) affect the balance of T-cell fates. In our current research, we are exploring the model's predictions when introducing a single mutated cell into the system. Which mutations have the potential to disrupt thymic homeostasis and lead to a tumour-like phenotype? Are these mutations in line with findings in patients of T-cell acute lymphoblastic leukemia (T-ALL)?
References:
- Bajoghli, B., Dick, A. M., Claasen, A., Doll, L., & Aghaallaei, N. (2019). Zebrafish and medaka: two teleost models of T-cell and thymic development. International Journal of Molecular Sciences, 20(17), 4179. [link]
- Aghaallaei, N., Dick, A. M., Tsingos, E., Inoue, D., Hasel, E., Thumberger, T., ... & Bajoghli, B. (2021). αβ/γδ T cell lineage outcome is regulated by intrathymic cell localization and environmental signals. Science Advances, 7(29), eabg3613. [link]


